The Sheba Pandemic Preparedness Research Institute
About SPRI
Our MODEL
SPRI ACTIVITIES
COLLABORATION
LEADERSHIP
PUBLICATIONS
Recent News
About SPRI
As COVID-19 began to spread in January 2020, Sheba’s Infection Prevention and Control Unit, headed by Prof. Regev-Yochay, was tasked with leading response efforts at Sheba. After recruiting over 9,000 healthcare workers, Sheba ran long-term studies that provided insight into real-world vaccine effectiveness in preventing asymptomatic and symptomatic infections, the immune response, healthy vs. vulnerable populations, and much more.
As a result of this work, over 60 research articles were published in renowned medical journals, such as the New England Journal of Medicine, the Lancet, Nature, and JAMA – impacting national decision-making and prompting the Israeli Ministry of Health, for instance, to administer a third booster shot.
Based on its experience during the COVID-19 pandemic, Sheba recognized the need to prepare for future pandemics and established the Sheba Pandemic Preparedness Research Institute (SPRI).
SPRI develops the medical advances and technologies needed to respond rapidly to global epidemiological crises, focusing on appropriate preventive measures and biological countermeasures such as fast diagnostic tests, monoclonal antibodies (passive immunization), vaccines, and more.

Prof. Gili Regev-Yochay
Director of SPRI
The SPRI Model
SPRI features a multidisciplinary team of virologists, immunologists, epidemiologists, clinicians, bioinformatics scientists, FACS experts, clinical study coordinators, and more. It aims to enable a continuum from basic science into products that will be tested pre-clinically, and then in Phase I studies at Sheba, alongside industry collaborations and partnerships that will lead to therapeutics and vaccines.
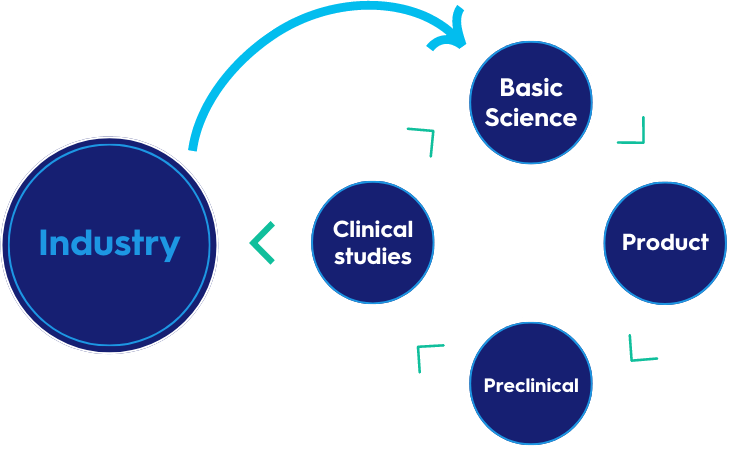
SPRI Activities
- Establishing multidisciplinary, multifaceted and collaborative research projects to translate clinical expertise into products for medical use.
- Deploying biological countermeasures promptly in the event of epidemic & pandemic threats.
- Ensuring pandemic preparedness through:
- Clinical research on emerging pathogens and host responses
- Developing vaccines at our Vaccine Research Center (VRC)
- Advancing diagnostic innovations related to emerging pathogens
- Modeling epidemiologic and mathematical steady-state and outbreak dynamics
International Collaborations

US National Institutes of Health Partnership
As part of efforts to turn research discoveries into life-saving solutions, Sheba signed a memorandum of understanding with the Dale and Betty Bumpers Vaccine Research Center (VRC) at the US National Institutes of Health (NIH).
Together, Sheba and the NIH-VRC will apply scientific and technological knowledge to design and develop vaccines and biologics against infectious diseases, combining research and clinical fields.
Additional International Collaborations
- Harvard School of Public Health
- Doherty Institute for Infection and Immunity
- Marc Lipsitch Harvard School of Public Health
- Simon Cauchemez, Pasteur Institute
SPRI Leadership

Prof. Gili Regev-Yochay
SPRI Director

Dr. Daniel Douek
SPRI Senior Scientific Advisor

Dr. Yaniv Lustig
SPRI Chief Virologist
Recognized nationally and internationally as an authority in his field, Dr. Lustig Serves as Director of the Israeli Ministry of Health’s Central Virology Laboratory, as well as SPRI Chief Virologist.

Dr. Yael Ottolenghi (Weiss)
Head of the SPRI Laboratory
With a Ph.D. in Immunology from Tel Aviv University, an MSc in Biotechnology from the Hebrew University, and postdoctoral immunology training at Tel Aviv University, Dr. Ottolenghi is a recognized national leader in her field.

DR. ABRAHAM MITTELMAN
Consultant/Senior Advisor to Vaccine Research Center (VRC)
Serving as a Consultant/Senior Advisor at the Vaccine Research Center (VRC), National Institute of Allergy and Infectious Diseases, Dr. Abraham Mittelman is a renowned authority in his domain. He acquired a Master of Public Health from Johns Hopkins University and a Master of Arts from Yeshiva University. His academic journey includes a specialized graduate program at the Massachusetts Institute of Technology (MIT), culminating in a Certificate in Toxicology. Dr. Mittelman further lends his expertise in operations and management as an advisor at SPRI.

Dr. Noam Barda
SPRI Chief Epidemiologist
With a BSc in Computer Science, MSc in Biostatistics, Ph.D. in Epidemiology and Public Health, an MD from Tel Aviv University, and postdoctoral training at Harvard Medical School, Dr. Barda serves as SPRI Chief Epidemiologist as well as Head of the Real World Evidence Research and Innovation Lab at the ARC Innovation Center.

Dr. Gili Joseph
SPRI Clinical and Epidemiological Research Manager
Dr. Joseph holds a Ph.D. in Immunology as well as an M.Sc. in Physiology and Pharmacology from Tel-Aviv University. She previously worked as a postdoctoral fellow at Rutgers University.

Shiri Reznik
SPRI Administrative Manager
Shiri Reznik expertly manages the extensive logistical and administrative requirements at SPRI, playing a pivotal role in advancing its scientific objectives. She earned her MHA in System Management and Public Health from Bar-Ilan University. Previously, Shiri served as Operations Manager at Clalit Health Services and as Administrative Manager at “Maccabi Tivee,” an innovative alternative clinic.
Recent Publications:
Correlates of protection against COVID-19 infection and intensity of symptomatic disease in vaccinated individuals exposed to SARS-CoV-2 in households in Israel (ICOFS): a prospective cohort study
Immunogenicity of Omicron BA.1-adapted BNT162b2 vaccines: randomized trial, 3-month follow-up
The Use of Rapid COVID-19 Antigen Test in the Emergency Department as a Decision-Support Tool
Reduced Neutralization Efficacy against Omicron Variant after Third Boost of BNT162b2 Vaccine among Liver Transplant Recipients
Immunogenicity and efficacy of fourth BNT162b2 and mRNA1273 COVID-19 vaccine doses; three months follow-up
High Immune Response Rate to the Fourth Boost of the BNT162b2 Vaccine against the Omicron Variants of Concern among Liver Transplant Recipients
Immunogenicity and efficacy of fourth BNT162b2 and mRNA1273 COVID-19 vaccine doses; three months follow-up
Recent News
Contact Us





